Drug delivery is changing - and the evidence is now visible in the regulatory record. Demographic shifts, scientific breakthroughs, and supply chain pressures are collectively reshaping which drug formats reach patients, how they are administered, and what packaging they require. For companies across the pharmaceutical value chain, the strategic implications are significant and time-sensitive.
Reading the Approval Data Differently
Most market analyses of drug delivery trends rely on sales volumes or R&D pipeline data. A more revealing approach is to examine drug approval data across multiple time horizons simultaneously - comparing all-time approval shares against the last decade, the last three years, and the last twelve months. Applied to regulatory data from the FDA and Germany's Bundesinstitut für Arzneimittel und Medizinprodukte, this methodology makes it possible to distinguish durable structural shifts from short-term fluctuations with a degree of empirical confidence that forward-looking projections rarely achieve.
The findings were cross-validated against interviews with practitioners spanning pharma consulting, clinical medicine, and pharmaceutical R&D. Their perspectives and the approval data point in the same direction.
What Experts Identify as the Defining Trends
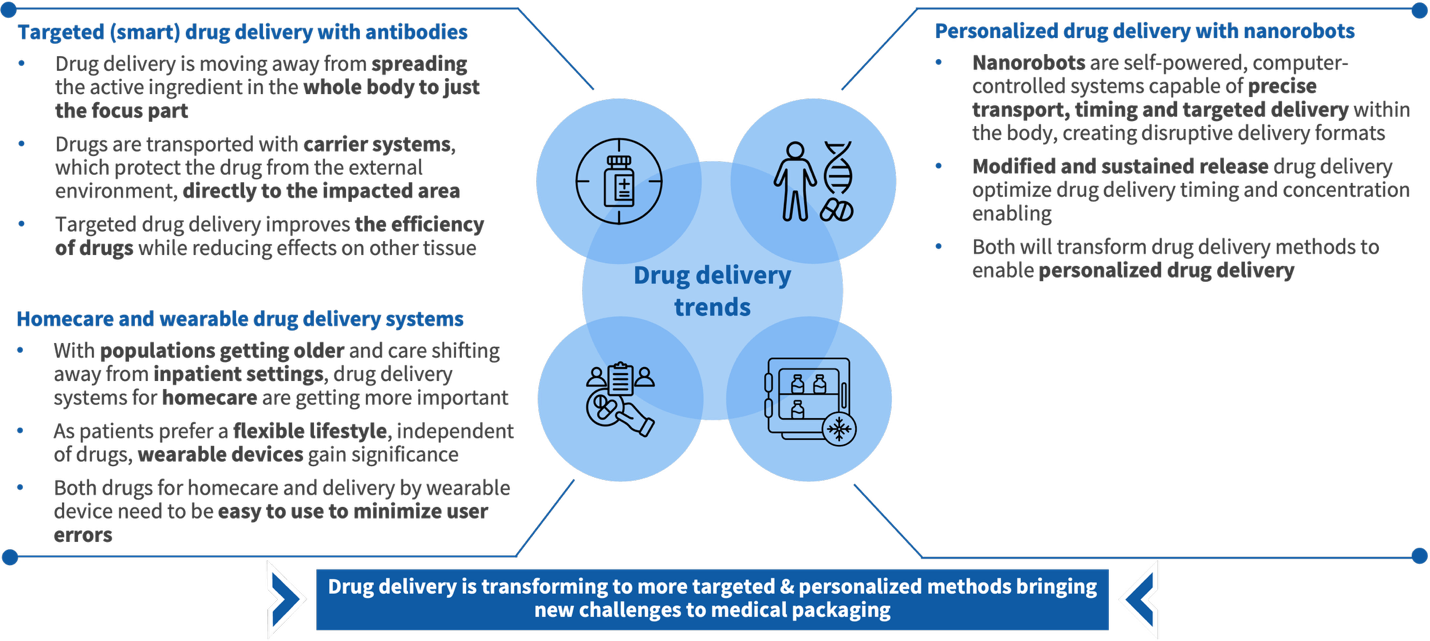
Across all interviews, three forces emerge as the dominant drivers of change in pharmaceutical drug delivery: the rise of homecare, the shift toward personalized medicine, and the advancement of targeted drug delivery. These are not emerging themes - practitioners describe them as already reshaping R&D priorities, clinical practice, and packaging requirements.
Four specific trends translate these forces into concrete format implications.
Targeted drug delivery with antibodies is moving the industry away from systemic distribution toward precision delivery to affected tissue only. Carrier systems protect compounds from the external environment and transport them directly to the impacted area - improving therapeutic efficacy while reducing adverse effects. This shift is a primary driver of growing approval share for injections and infusions.
Personalized drug delivery via nanorobotics extends this logic further. Self-powered, computer-controlled systems enabling precise, sustained-release delivery at the cellular level are moving from research into early commercial application, transforming drug delivery from a standardized intervention into an individualized therapeutic event.
Homecare and wearable drug delivery is becoming a structural necessity as aging populations shift care out of clinical settings. Wearable delivery devices are gaining commercial traction, but their viability hinges on ease of use - packaging design and delivery simplicity are becoming competitive differentiators, not afterthoughts.
Ultra-deep freeze drug delivery, accelerated by mRNA vaccine technology, presents perhaps the most immediate operational challenge. Therapies requiring storage at temperatures as low as -70C create new demands for primary and secondary packaging. Silicone is emerging as the preferred material for its low-temperature stability, but cold-chain infrastructure, packaging specifications, and Track & Trace compliance requirements represent a new operational frontier for most industry participants.
What the Approval Data Shows
The regulatory data validates each of these expert perspectives - and does so consistently across two independent markets.
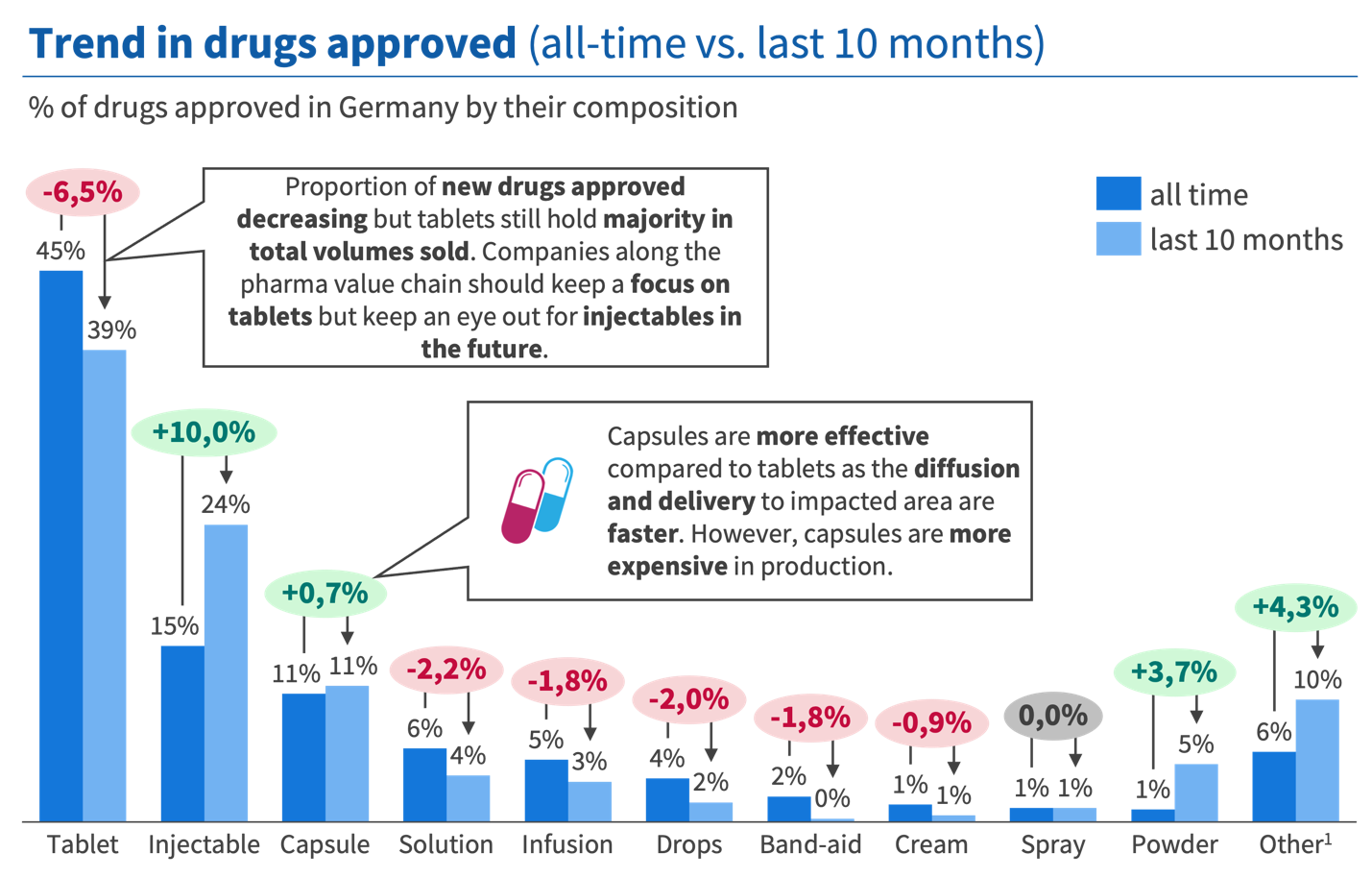
In Germany, tablets remain the highest-volume format, but their share of newly approved drugs has declined 6.5 percentage points in recent approvals, settling at 39%. Injectables have surged by 10 percentage points. Powder is up 3.7 points. Capsules are gaining ground, reflecting their faster diffusion and more targeted delivery relative to traditional tablets, despite higher production costs.
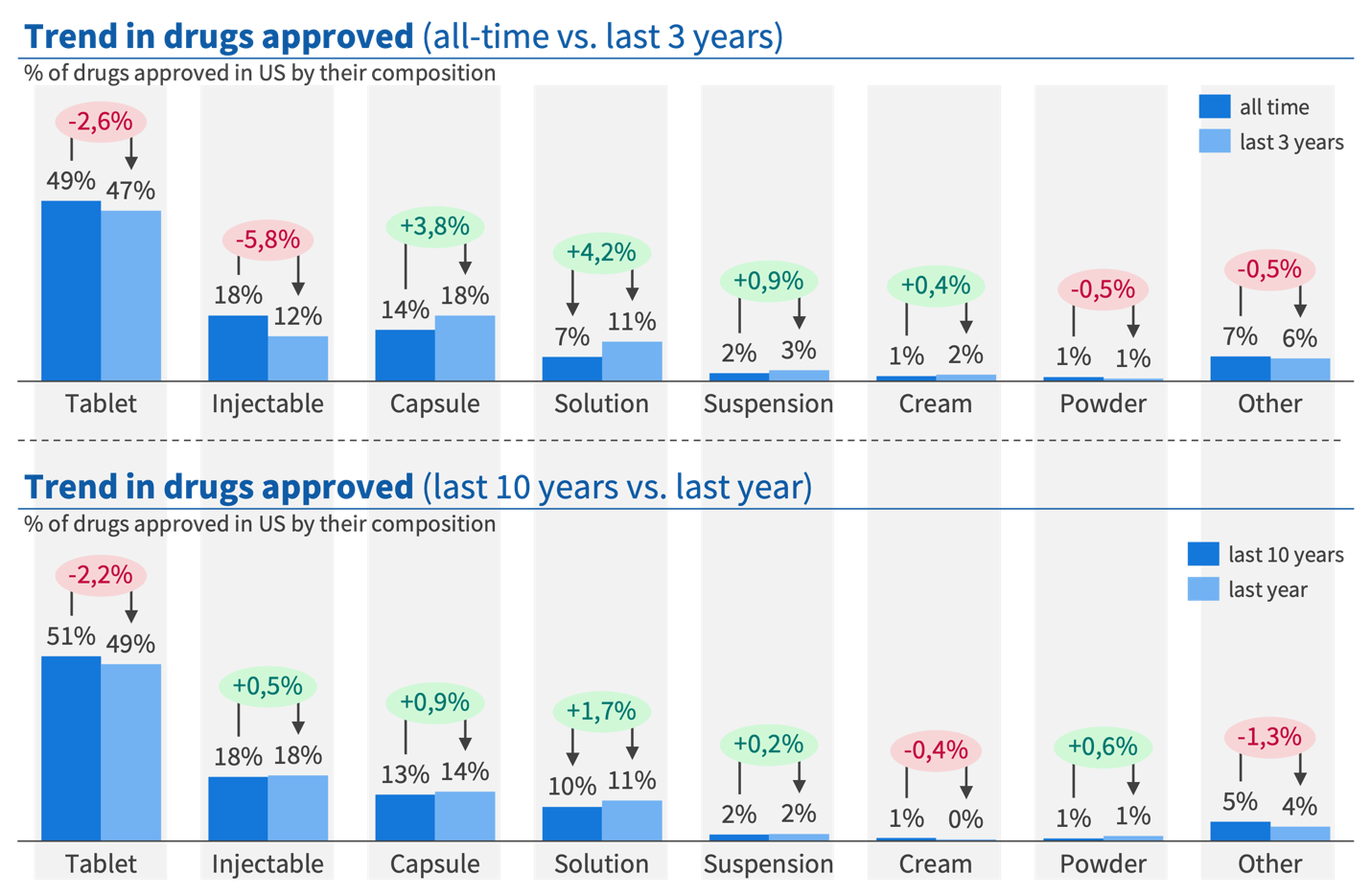
The United States shows the same pattern. Tablets account for 47-49% of all-time approvals but are declining by 2.6 percentage points when recent years are compared against the historical average. Capsules are up 3.8 percentage points, solutions 4.2 points, and suspensions are rising. Examining delivery type rather than composition reinforces the picture: intravenous delivery is up 5.2 percentage points and subcutaneous up 1.6 points over recent years, directly reflecting the targeted delivery trend in real approval data.
The same directional signals appearing independently across two major markets, replicated across multiple time horizons, are a strong indicator of structural rather than cyclical change.
Three Implications for Industry Participants
Tablets remain the volume foundation - but should not distract from what is coming. Tablets still command the majority of drug volumes across developed and developing markets. Prematurely pivoting away from this segment would be a strategic mistake. At the same time, treating tablet dominance as a reason to delay investment in adjacent formats is equally risky.
The near-term growth opportunity lies in injections, infusions, and capsules. Approval data and expert perspectives converge on a decisive shift toward these formats in developed markets. Companies that build operational capability now - before the demand inflection becomes consensus - will establish positions that are difficult for later movers to replicate.
Ultra-deep freeze and complex molecule packaging is the high-margin frontier of the next decade. As mRNA-based therapies expand from vaccines into oncology and rare diseases, cold-chain integrity, silicone material expertise, and Track & Trace compliance will define competitive differentiation. The window to build ahead of mainstream demand is narrowing, and the revenue per product in this segment is materially higher than in traditional formats.
The Strategic Takeaway
Drug delivery is bifurcating. Tablets will remain the commercial backbone of global pharmaceutical volumes for years to come, particularly in developing markets. Simultaneously, the innovation frontier is advancing toward targeted, personalized, and homecare-compatible mechanisms that carry fundamentally different packaging and logistics requirements. Companies that can serve both realities - with dedicated capability-building for each - are best positioned to capture disproportionate value in the years ahead.
Sources: Raumedic insights (2023), ASC Publications (2023), Journal of Multidisciplinary Healthcare (2021), Bundesinstitut für Arzneimittel und Medizinprodukte, FDA (Drugs@FDA 2024), icons analysis.
